Further reading
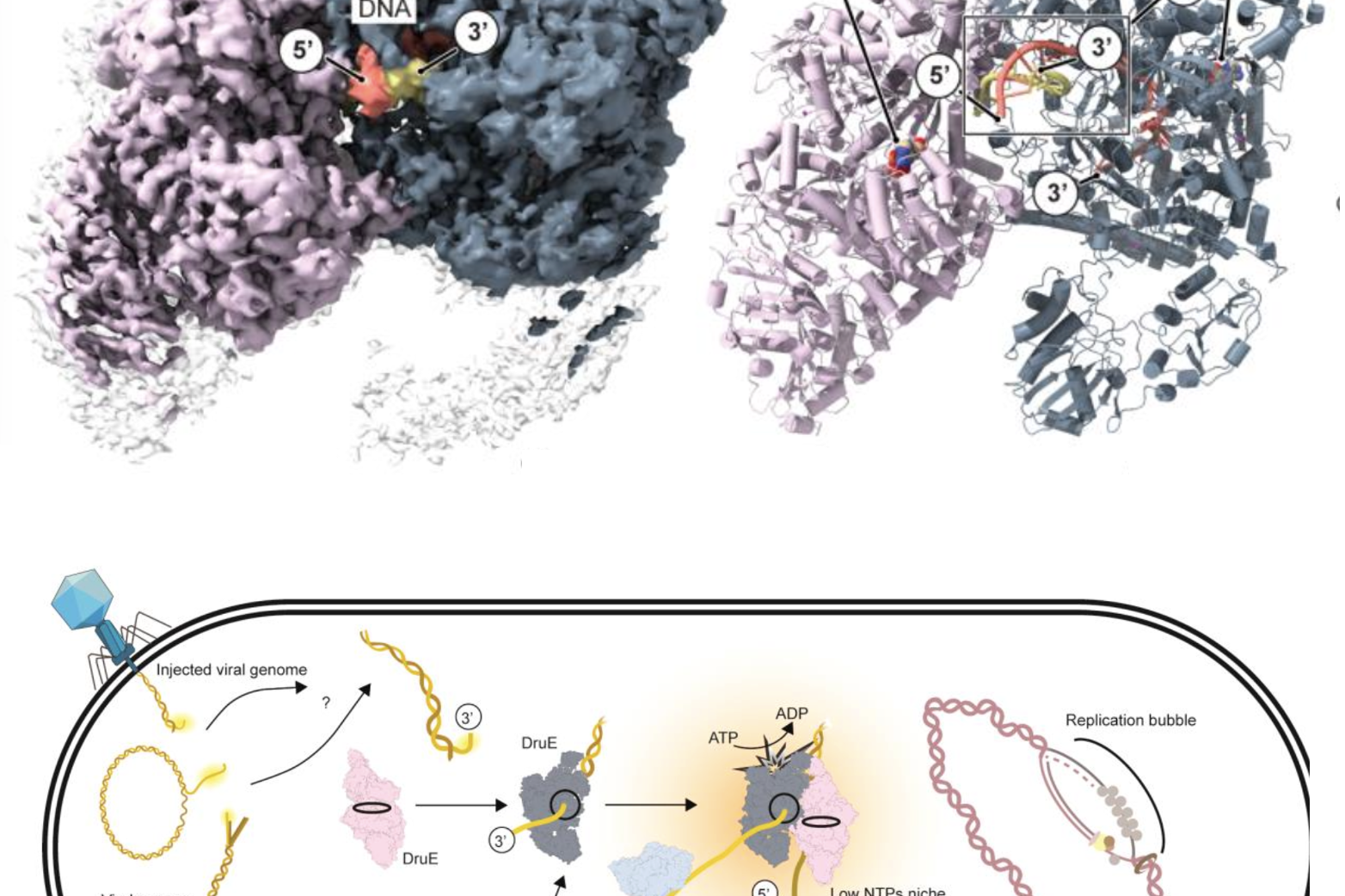
Molecular basis of the Druantia anti-phage defense system – out now on bioRxiv
Congratulations to Haidai and everyone involved in this project!
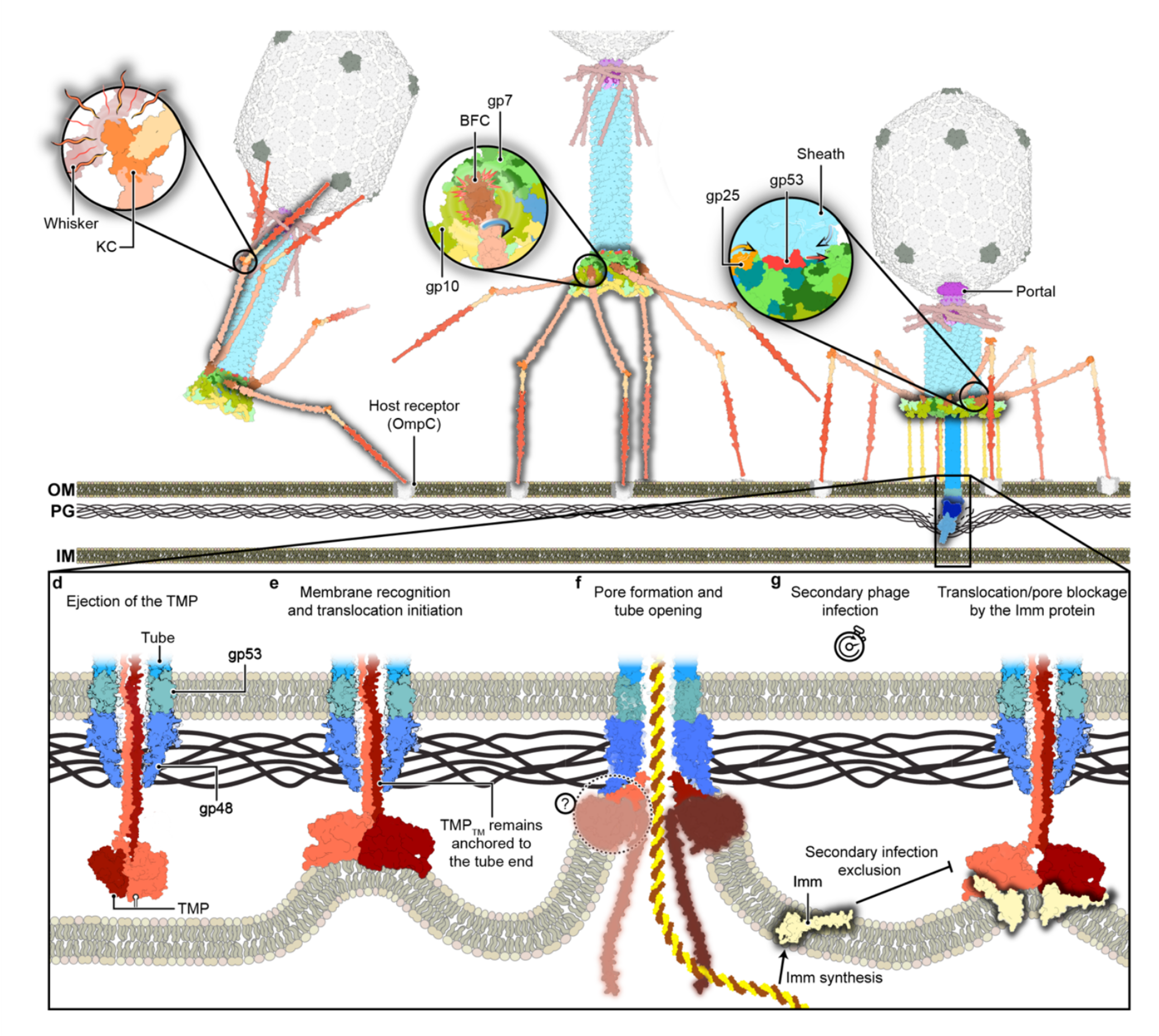
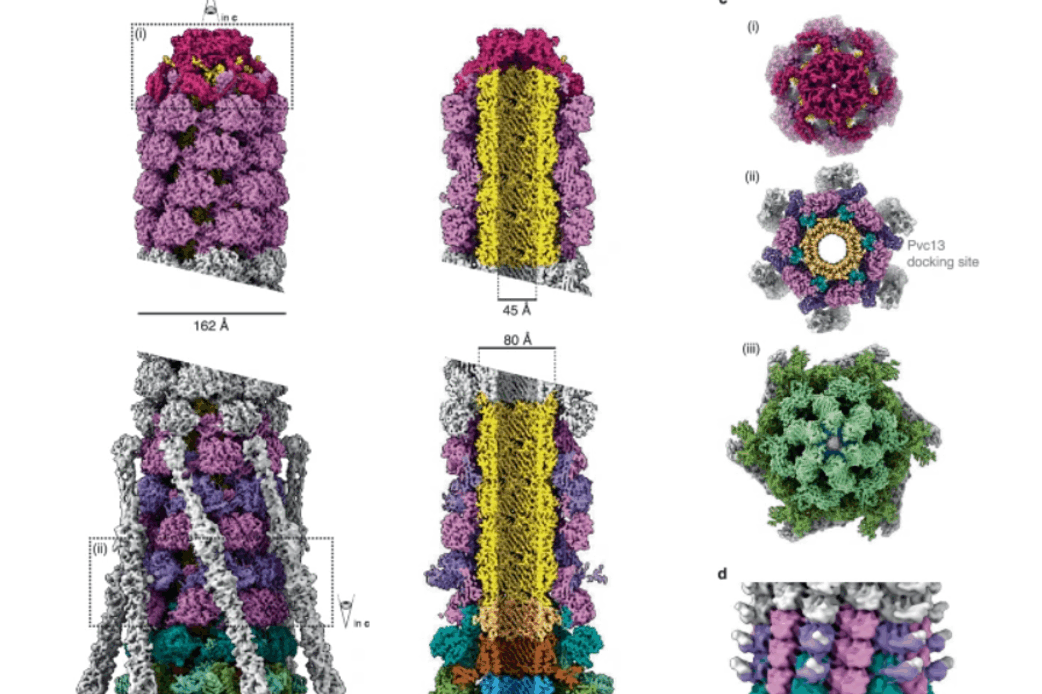
Extracellular contractile injection system from Photorhabdus luminescens structure paper out now in Nature Communications
Congratulations to Leyre and everyone involved in this project!
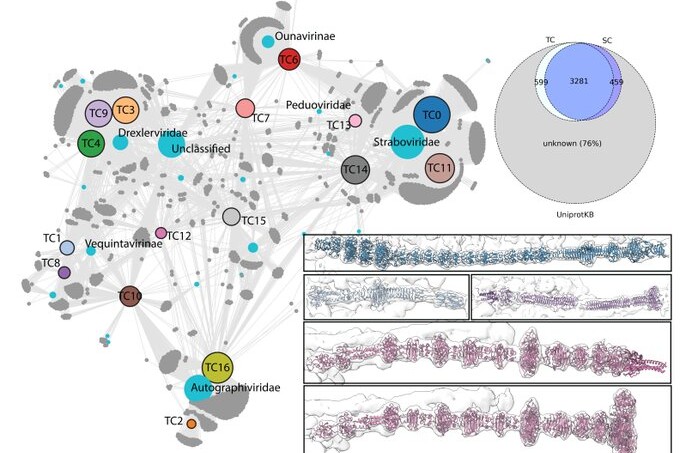
RBPseg out now in Science Advances
Congratulations to Victor and everyone involved in this project!